
Most of the space in an atom is empty as the particles passed through the gold foil without any hindrance.He observed the trajectory of the alpha (α)-particles after passing through an atom and drafted some postulates of the experiment, which are: Rutherford conducted an experiment bombarding the alpha (α)-particles on a gold foil. Mentioned below are the important theories about the structure of an atom as per the chapter.Ĭredits: Aasoka Rutherford’s Model of an Atom 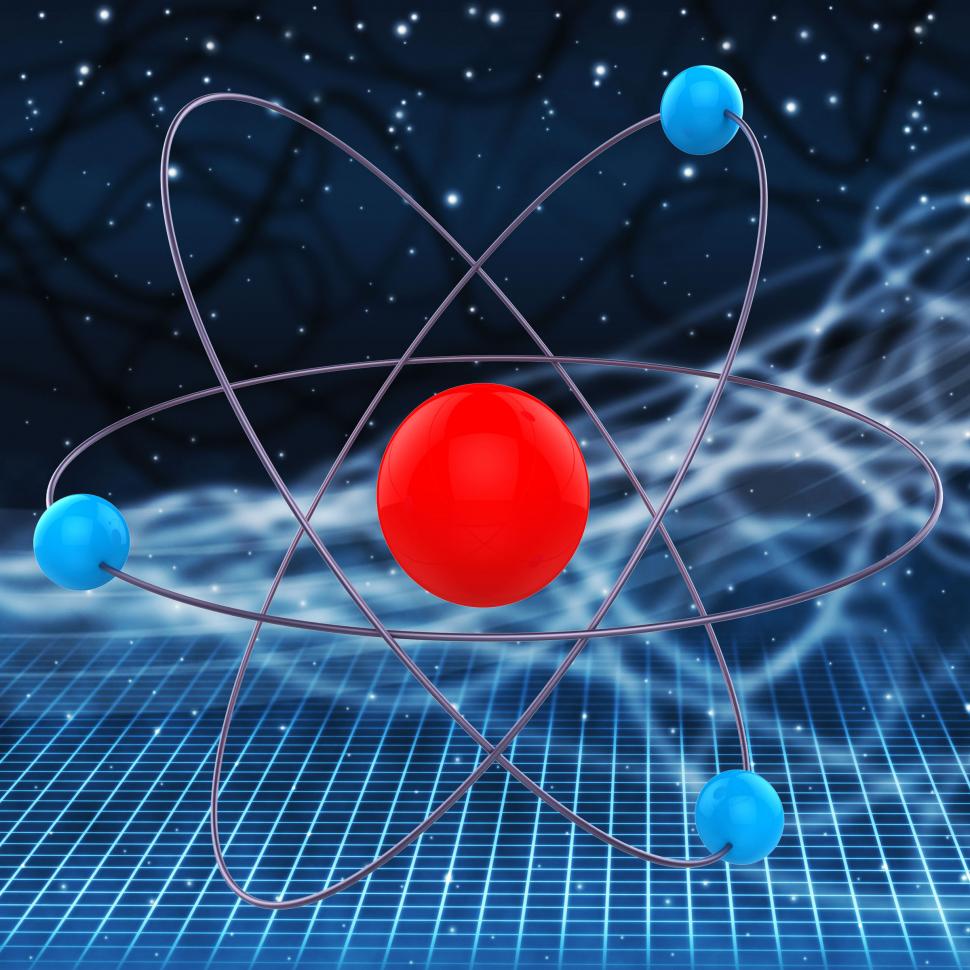
Since the time of the discovery of atoms, there are a variety of theories which were formulated by many renowned scientists. The Fundamental Unit Of Life Class 9 Notes Different Models on Structure of an Atom Helium atom contains two protons, two electrons and two neutrons making its mass number at 2. The hydrogen atom (H) contains only one proton, one electron, and no neutrons. The oxygen atom contains eight protons, eight electrons and eight neutrons making its mass number at 16. The carbon atom contains six protons, six electrons and six neutrons making its mass number at 12. These neutrally charged particles are Neutrons. Chadwick discovered a subatomic particle with no charge and a mass equivalent to protons in the nucleus of all atoms. ProtonsĮrnest Goldstein, in 1886, discovered that with a different condition in the same chamber, anode emitted positively charged particles known as Canal rays or later named as Protons. These negatively charged particles are Electrons. Thomson, in 1897, discovered negatively charged particles emitted by the cathode towards the anode in a cathode ray experiment. 
The electrons are located at the outermost regions called the electron shell. The nucleus of the atom contains protons and neutrons where protons are positively charged and neutrons are neutral. Class 9 ICSE Maths Fundamental Constituents of an AtomĪn atom contains three basic particles namely protons, neutrons and electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
